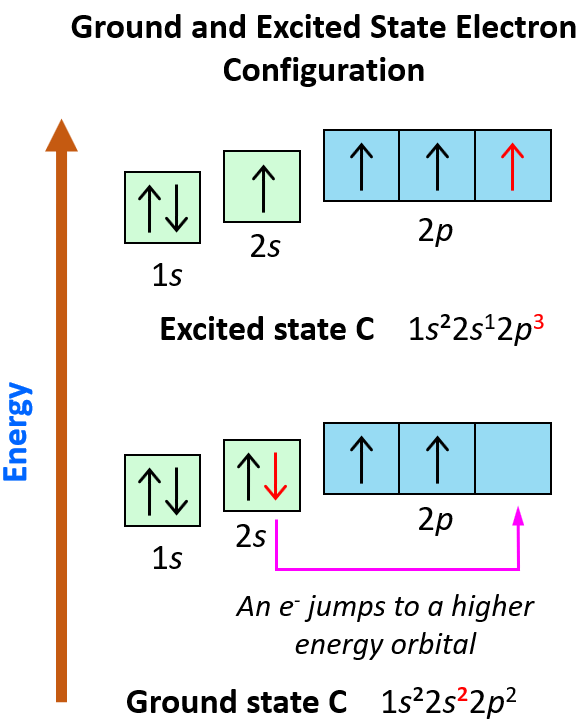
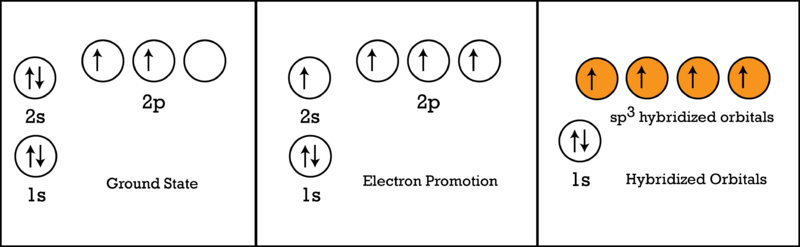
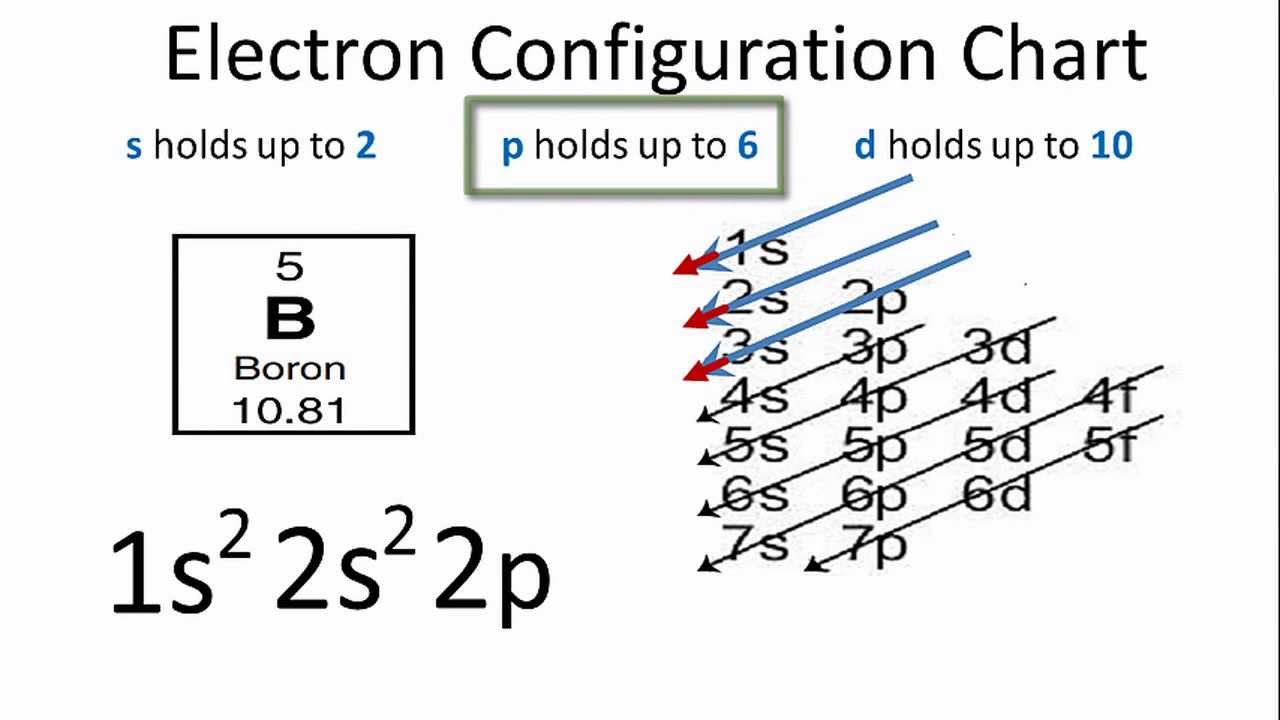
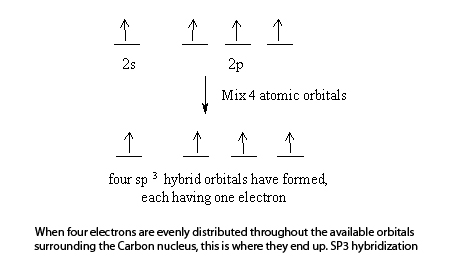
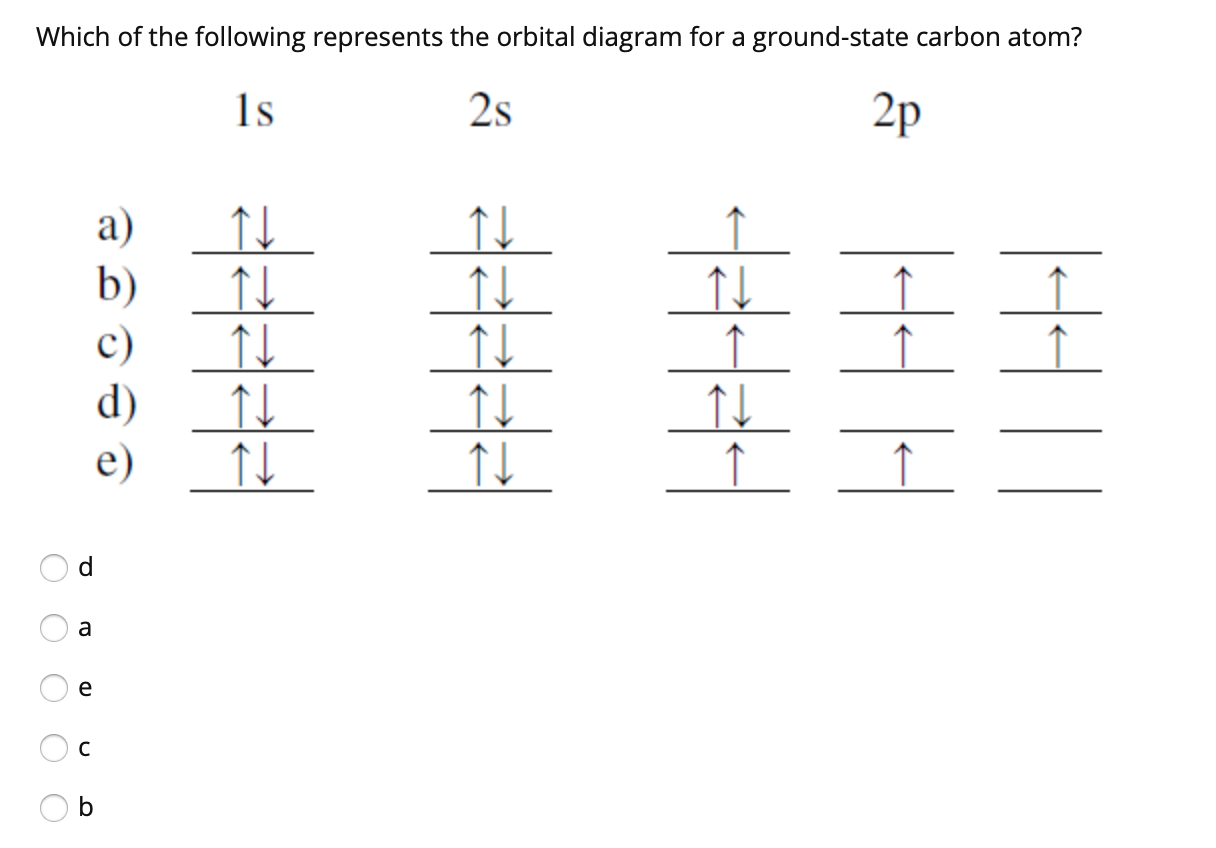
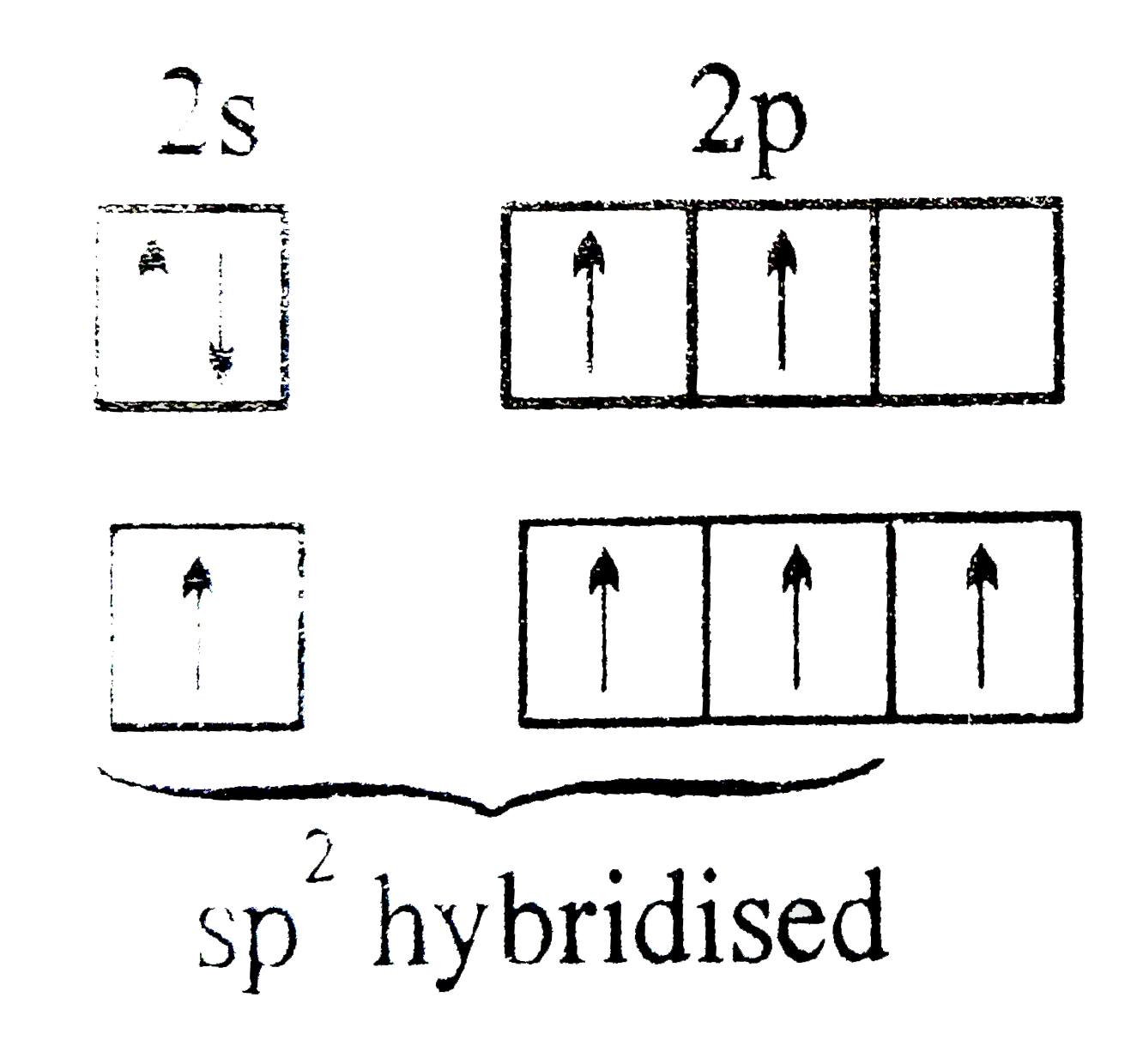Explain the four unpaired electrons in carbon atom through excited state. - Sarthaks eConnect | Largest Online Education Community

1: Electronic configurations for carbon in the ground state (left) and... | Download Scientific Diagram

Electronic configuration diagram vs energy for carbon atom in its (a)... | Download Scientific Diagram

Write the ground state electron configuration for a neutral carbon atom, and for an excited state of carbon? - CBSE Tuts
3: Electronic ground state (a), excited state (b) of the carbon atom,... | Download Scientific Diagram

Explain the four unpaired electrons in carbon atom through excited state - CBSE Class 10 Science - Learn CBSE Forum
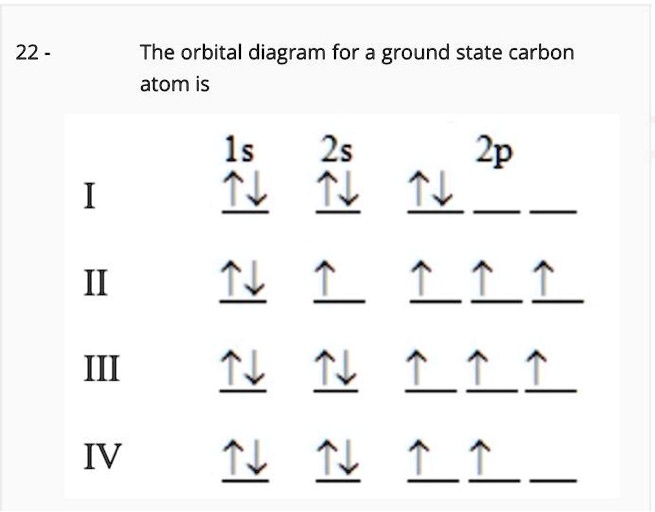
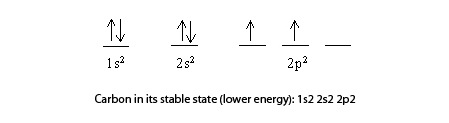
SOLVED: The orbital diagram for a ground state carbon atom is 22 1s 2s 2p 1 ^ 4 1 77 1 ^ 77 1 ^ LL IV