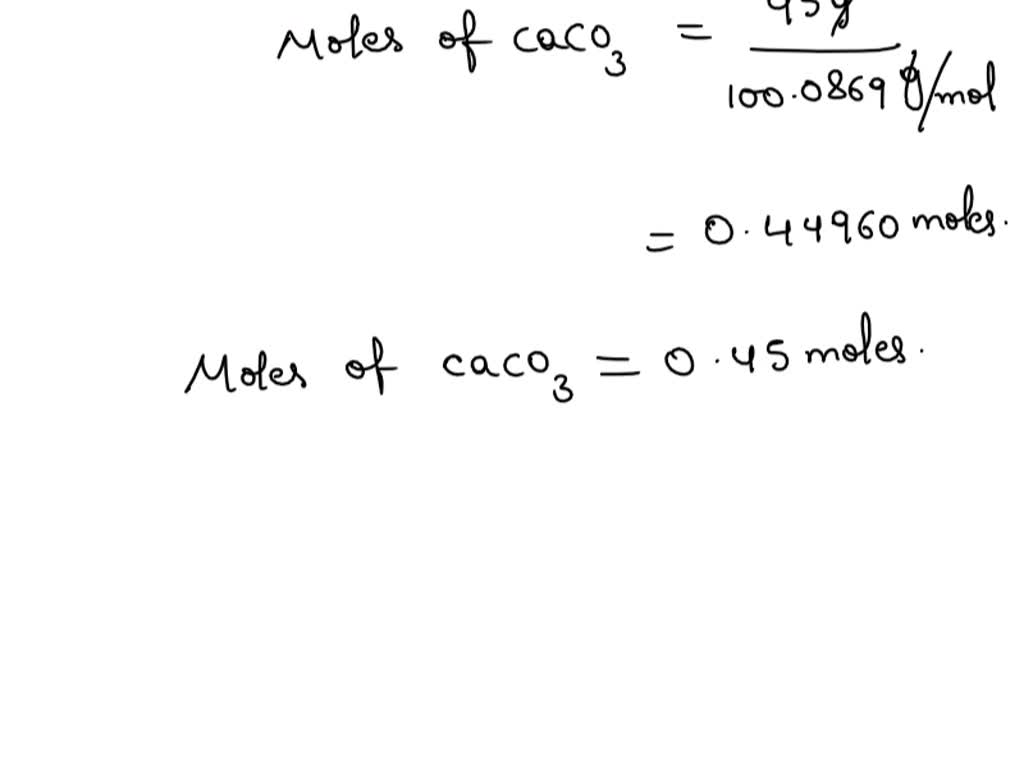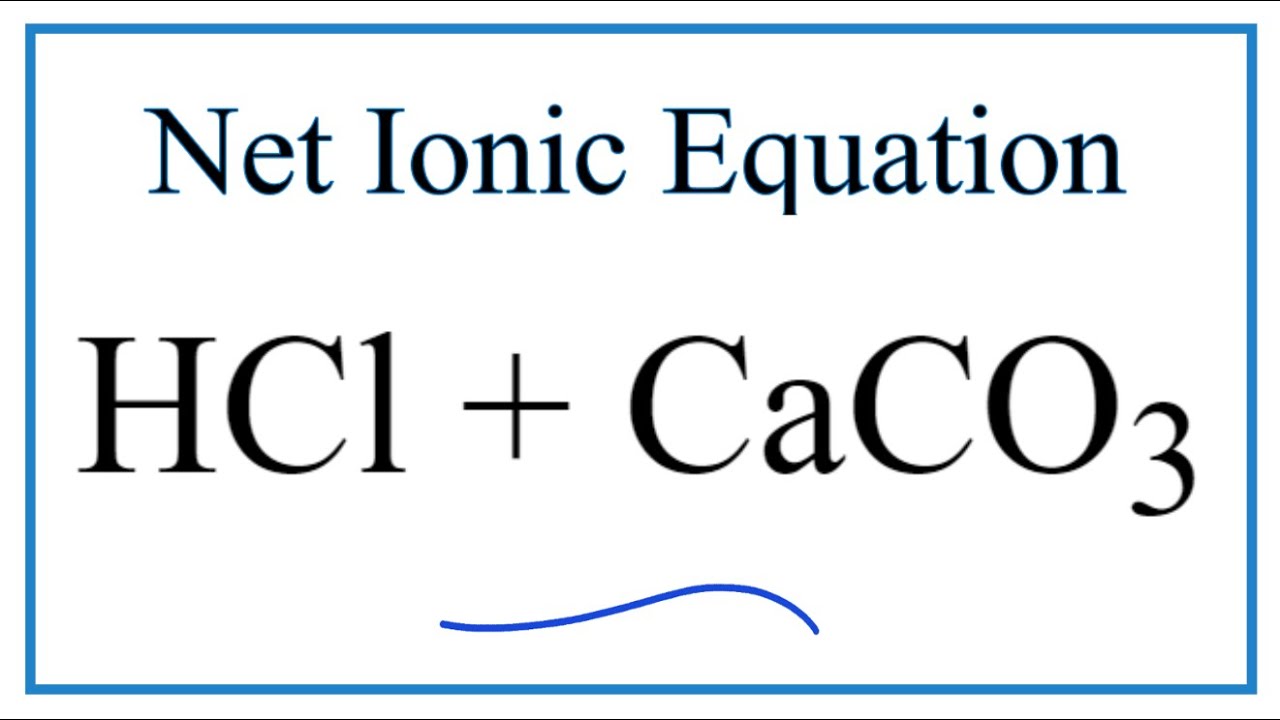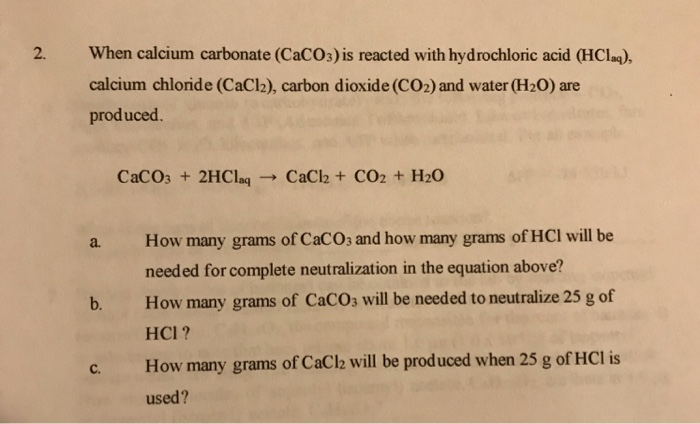Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

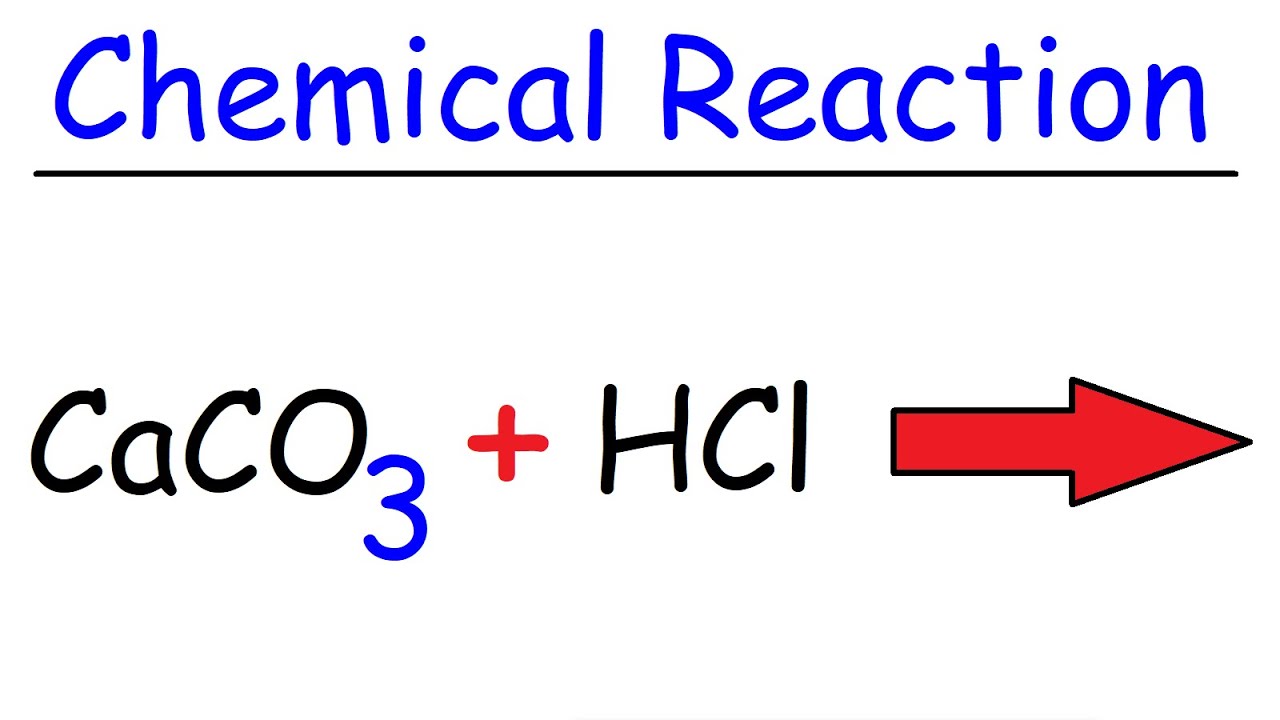
CaCO3+HCl=CaCl2+CO2+H2O Balanced Equation||Calcium Carbonate+Hydrochloric acid Balanced Equation - YouTube

Balance the following equations : (a) Caco, (s) + HCl (aq) + CaCl, (aq) + H2O (1) + CO, (g) (b) Zn (s) + HCl - Brainly.in

Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) .What mass of CaCO3

Calcium carbonate reacts with aqueous `HCl` to give `CaCl_(2)` and `CO_(2)` according to the rea... - YouTube
What is the pH reading after the endpoint of the neutralization reaction of calcium carbonate and HCL? - Quora